
If it gains an electron, it becomes a negative ion. If a neutral atom loses an electron, it becomes a positive ion. 
Ordinary atoms that either gain or lose electrons are called ions. Some atoms, however, lose or gain electrons in chemical reactions or in collisions with other particles. Thus the positive and negative charges are balanced. If it did not have six protons, it would not be carbon.Īn ordinary atom has an equal number of protons and electrons. Carbon-14 is an isotope with eight neutrons per atom. Atoms concurrency model eliminates requirements for mutual exclusion algorithms. It features compile time task scheduling and produces code with improved memory usage and execution time. Carbon-12, the ordinary form of carbon, has six protons and six neutrons per atom. Atom is a concurrent and domain specific programming language that is used for real-time and embedded applications. Atoms are responsible for giving each matter its. For example, for carbon, the mass number is 12, and the atomic weight is 12.011.Ītoms that have the same atomic number but different mass numbers are called isotopes. The matter is composed of invisible, indivisible, innumerable particles called atoms. The mass number and atomic weight are very similar. The atomic weight is equal to the mass number divided by a certain number that scientists have come up with. The mass number is equal to the total number of protons and neutrons in an atom. Two other related properties of atoms are the mass number and the atomic weight. No other element has an atomic number of 1. In electrically neutral atoms, the number of protons equals the number of electrons. For instance, hydrogen has an atomic number of 1 because every hydrogen atom has one proton in its nucleus. atom t m The smallest unit of an element, consisting of at least one proton and (for all elements except hydrogen) one or more neutrons in a dense central nucleus, surrounded by one or more shells of electrons. An atom is the smallest unit of an element, and each chemical element has a unique atomic number. Every atom is associated with a specific chemical element. The atomic number determines what kind of atom it is. Join Seneca to get 250+ free exam board specfic A Level, GCSE, KS3 & KS2 online courses.The single most important thing to know about an atom is how many protons it has in its nucleus. : an elementary particle that is identical with the nucleus of the hydrogen atom, that along with the neutron is a constituent of all other atomic nuclei, that carries a positive charge numerically equal to the charge of an electron, and that has a mass of 1. 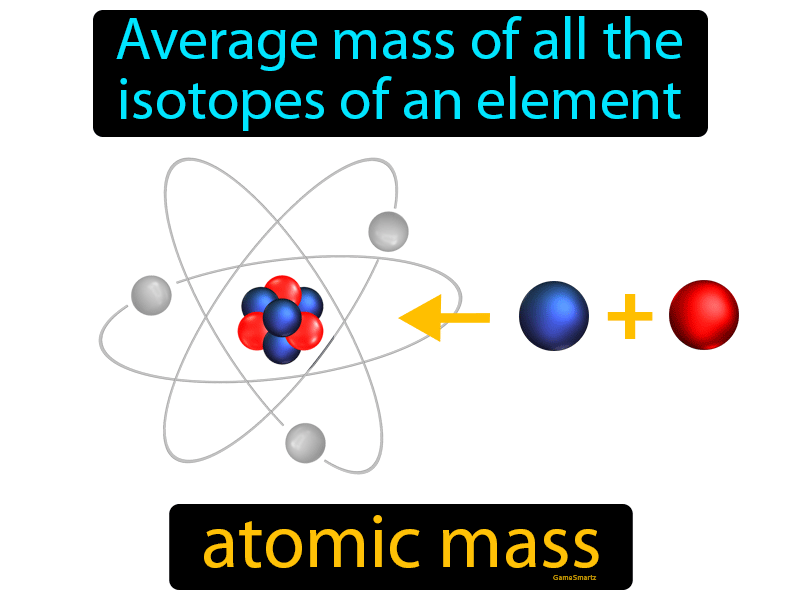
Want to learn more about Atom Size & Number? This information can be found for each element in the periodic table. The atomic number of an atom is the number of protons in the atom. The mass number of an atom (also called relative mass) is the total number of protons and neutrons in a nucleus. Relative mass of electrons = 0.0005 (this gets rounded to zero).Ītoms: Relative mass = number of protons + number of neutrons. **Mass: **Sub-atomic particles: Relative mass of protons and neutrons = 1. 
If an atom was the size of Wembley Stadium, then the nucleus would be the size of a garden pea. The nucleus of an atom is 10,000 times smaller than the atom. About 5 million hydrogen atoms could fit into a pin head. **Size: **Atoms have an average radius of about 0.1 nm. They are made up of sub-atomic particles (protons, neutrons, and electrons), which are even smaller and lighter than an atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
